Coronavirus April 2021—Part 12 Should You Get the Johnson & Johnson Vaccine?
In this post, we answer your questions about the Johnson & Johnson vaccine, which was recently authorized by the FDA for distribution. As usual, if you’re less interested in how we got to our conclusions than you are in the conclusions themselves, feel free to skip to the BOTTOM LINE in each section and the CONCLUSION at the end.
Question: Should you get the vaccine when it becomes available to you?
Answer: Yes.
Nuts and bolts of the johnson & johnson vaccine
J & J’s COVID-19 vaccine has been dubbed Ad26.COV2.S. Unlike the Pfizer and Moderna vaccines, it’s a DNA vaccine. DNA is delivered in a special type of cold virus, an adenovirus, that’s “replication-incompetent,” meaning it doesn’t go on to infect other cells and make you sick. Instead, it codes for a modified version of the SARS-CoV-2 spike protein, the part of the SARS-CoV-2 virus that enables it to latch onto and enter human cells. The muscle cells into which the vaccine is injected begin manufacturing this modified spike protein and present it on their cell membranes, where the body’s immune system recognizes it and begins manufacturing antibodies against it.
The vaccine regimen requires only one dose (though a regimen that includes a second dose—a booster shot—is ongoing). When stored at 2º to 8º C, the vaccine has a shelf life of 3 months. It’s given intramuscularly (in the shoulder muscle). The vaccine is preservative-free.
Importantly, the analysis of the J & J trial has thus far only included subjects 18 years of age and older.
BOTTOM LINE: Like both the Moderna and Pfizer studies, the J & J study included subjects representative of the general population.
How are Vaccines Proven to be safe?
As we discussed in previous posts about COVID-19, vaccines are studied in three phases, each subsequent phase recruiting more subjects than the previous phases to test vaccine efficacy and safety. These studies are randomized, double-blind, and placebo-controlled—the gold-standard study design—because only randomized, controlled trials can definitively prove an intervention works and that it’s safe.
It turns out that, historically, most adverse reactions to vaccines occur within the first two months of vaccination. This is why J & J, like Pfizer and Moderna, applied for Emergency Use Authorization once most of the subjects in its study had been observed for 2 months after receiving the vaccine (the study is planned to go out 2 years before concluding). Post-marketing studies—long-term surveillance efforts looking for adverse events after a vaccine has been given to millions of people—have identified adverse events not seen in initial vaccine studies, but these events are, almost by definition, rare given the large numbers of subjects tested in Phase III trials (for example, Guillain-Barre syndrome occurring after a flu shot at a rate of literally 1 in a million). While questions have been raised about some vaccines causing, among other things, sudden infant death syndrome, pediatric asthma, autism, inflammatory bowel disease, and permanent brain damage, in each case, studies have revealed these claims to be groundless.
Other adenovirus-based vaccines like Johnson & Johnson’s COVID-19 vaccine have been used to vaccinate almost 200,000 people in studies and vaccination programs against diseases like Ebola, Zika, HIV, HPV, malaria, and respiratory syncytial virus. They’re considered safe.
What have been the adverse events in the J & J study? The most common were injection site pain at 48.6 percent (compared to 88.2 percent for Moderna and 84.1 percent for Pfizer), fatigue at 38.2 percent (compared to 65.3 percent for Moderna and 62.9 percent for Pfizer), headache at 38.9 percent (compared to 58.6 percent for Moderna and 55.1 percent for Pfizer), muscle pain at 33.2 percent (compared to 58 percent for Moderna and 38.3 percent for Pfizer), chills at 2 percent (compared to 44.2 percent for Moderna and 31.9 percent for Pfizer), fever at 9 percent (compared to 15.5 percent for Moderna and 14.2 percent for Pfizer). The frequency of serious adverse events was 0.4 percent (compared to less than 0.1 percent for Moderna and less than 0.5 percent for Pfizer) and, importantly—as with both the Moderna and Pfizer vaccines—evenly divided between the vaccine group and the placebo group, meaning they weren’t likely attributed to the vaccine itself.
There were a total of seven COVID-19-related deaths in the study, all in the placebo group, all in South Africa. This isn’t a high enough death rate to prove the vaccine prevents death from COVID-19. Also, the study didn’t present enough data to determine if the vaccine is effective at preventing asymptomatic infection.
Bottom line: Like both the Pfizer and Moderna vaccines, the J & J vaccine is safe. Any adverse events not yet identified will almost certainly be rare.
How are Vaccines Proven to be Effective?
The J & J Phase III study randomized approximately 40,000 subjects to receive either the vaccine or placebo. They chose to include this may subjects because they needed a sample size large enough to yield a sufficient number of cases of COVID-19 to tell if there was a statistically significant difference in infection rates between subjects who got the vaccine and subjects who got the placebo, and to detect rarer adverse events from the vaccine. By the time J & J submitted their application to the FDA for Emergency Use Authorization, there had been 259 cases of COVID-19 in their study participants, a number similar to that in trials testing vaccines for other diseases.
J & J found that their vaccine overall has a 66.1 percent efficacy rate in preventing symptomatic COVID-19 (lower than both Pfizer’s and Moderna’s roughly 95 percent efficacy rates). Vaccine efficacy is defined as the reduction in risk that vaccination provides in a clinical trial (when conditions are as perfect as they’re ever going to be). So a 66.1 percent vaccine efficacy means vaccination reduces the baseline risk (which differs in different circumstances) of getting COVID-19 by 66.1 percent. So, for example, if your baseline risk of getting COVID-19 from, say, your infected spouse is 27.8 percent (the highest risk reported in a clinical trial that we’ve seen), the risk of getting COVID-19 from your infected spouse if you’ve been vaccinated would be 66.1 percent less, or 9.4 percent.
This is a good level of risk reduction for a vaccine. For comparison, flu shots have an efficacy of 40 to 60 percent. The polio vaccine has an efficacy of 99 to 100 percent. The hepatitis B vaccine has an efficacy of 80 to 100 percent.
There weren’t enough subjects with proven prior COVID-19 infection included in the study, so we don’t know if vaccination reduces the likelihood of re-infection. Pregnant women were excluded from the trial, as were women who were breastfeeding and subjects who were immunocompromised, so we can’t say what effect or risk the vaccine brings to those groups.
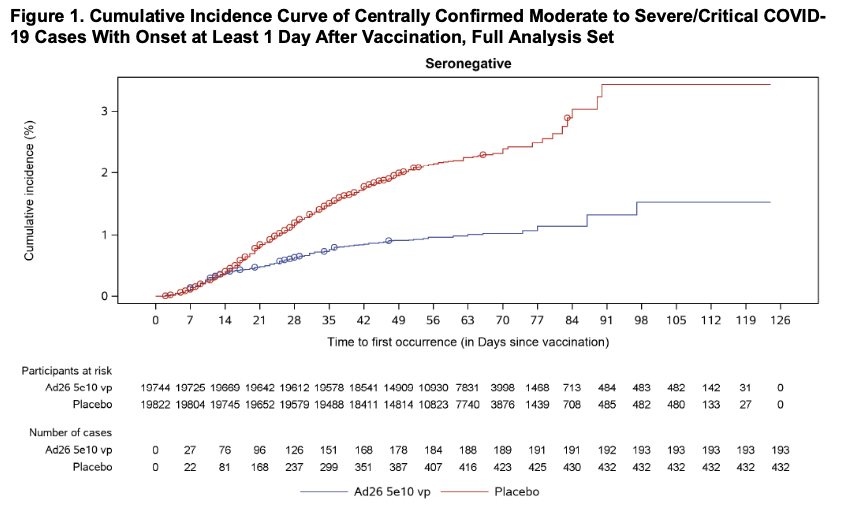
The graph below illustrates the cumulative incidence of COVID-19 infection between subjects in the vaccination group and subjects in the placebo group, which begins to diverge 14 days after the time of the vaccine injection:
Importantly, as with the Pfizer and Moderna vaccines, the J & J vaccine seems to be effective across age groups (above age 18), genders, racial and ethnic groups, and subjects with medical conditions associated with high risk for severe COVID-19. In this study, 39 subjects developed severe COVID-19, only 5 of whom received the vaccine, arguing for a 85.4 percent efficacy in preventing severe COVID-19.
Bottom line: J & J’s vaccine is effective for most people over the age of 18. It also appears that it prevents severe COVID-19, though perhaps not as effectively as Moderna’s vaccine. On the other hand, the variants now spreading in South Africa, Britain, and the U.S. weren’t circulating in the population when Moderna and Pfizer applied for Emergency Use Authorization. All the vaccines available so far prevent severe infection and hospitalization at a high rate, but not at 100% efficacy. This means even if you get vaccinated, there is a small chance you could still catch COVID-19, and an even smaller chance that if you do catch it that it will be a severe case. Given the size of the population, we’ll all hear about breakthrough cases. It’s important to realize this doesn’t mean the vaccine isn’t doing its job. And the vast majority of the small proportion of breakthrough cases will be mild to asymptomatic.
Unanswered Questions
- The study didn’t present enough data to prove the vaccine prevents asymptomatic infection. Nor did it assess whether subjects who developed COVID-19 despite vaccination are less likely to transmit the virus. Thus, it’s not yet clear how effective the vaccine will be in containing the spread of the infection. (A recent study from the CDC, however, strongly suggests that both mRNA vaccines—Pfizer’s and Moderna’s—do indeed prevent even asymptomatic COVID-19 infection by 90 percent in real-world circumstances, which is great news. We need more studies to learn if this is also the case for J & J’s vaccine.)
- The study hasn’t gone on long enough to tell if subjects who were vaccinated yet still contracted COVID-19 have a lower risk of long-term effects of COVID-19.
- We don’t yet know if the vaccine reduces the risk of dying from COVID-19 if you get COVID-19 despite being vaccinated (though, as with Moderna’s and Pfizer’s vaccines, if it prevents serious COVID-19 infection, presumably some of those infections it prevents would have led to death).
- There was insufficient data to draw conclusions about safety and efficacy of the vaccine in children younger than 18, pregnant or breastfeeding women, and patients who are immunocompromised.
- We don’t yet know how long immunity lasts and whether or not booster shots will be necessary.
Conclusions
- The vaccine is reasonably effective in preventing symptomatic COVID-19 infection.
- The vaccine is safe. Adverse reactions, both local and systemic, are mostly minor. Though the study hasn’t yet gone on long enough to prove there are no serious long-term adverse affects, such adverse affects, if they exist, are likely to be rare and non-life-threatening.
- We recommend everyone who is eligible to receive the vaccine should receive it when it becomes available to them.
- It very well may take all of 2021 to get everyone who’s willing to be vaccinated to receive the shots, which means it likely won’t be until early 2022 that life returns to pre-pandemic normal. In the meantime, continue to wear a mask when indoors with anyone you don’t live with, wash your hands frequently, and refrain from dining indoors at restaurants.
- Coronavirus February 2020—Part 1 What We Know So Far
- Coronavirus March 2020—Part 2 Measures to Protect Yourself
- Supporting Employee Health During the Coronavirus Pandemic
- Coronavirus March 2020—Part 3 Symptoms and Risks
- Coronavirus March 2020—Part 4 The Truth about Hydroxychloroquine
- Coronavirus April 2020—Part 5 The Real Risk of Death
- Coronavirus April 2020—Part 6 Evaluating Diagnostic Tests
- Coronavirus April 2020—Part 7 The Accuracy of Our Antibody Test
- Coronavirus May 2020—Part 8 How to Reopen a Business Safely
- Coronavirus August 2020—Part 9 Masks, Vaccines, and Rapid Testing
- Coronavirus December 2020—Part 10 Should You Get the Pfizer Vaccine?
- Coronavirus December 2020—Part 11 Should You Get the Moderna Vaccine?
[jetpack_subscription_form title=” subscribe_text=’Sign up to get notified when a new blog post has been published.’ subscribe_button=’Sign Me Up’ show_subscribers_total=’0′]

Well written – learned more in two minutes here than I could any other way.
Is the figure of 1/1,000,000 of GB Syndrome after a flu shot higher than expected without a flu shot, or with a shot that is not flu?
I hope J&J gets their production quality issues under control.
Re: storage temperature for the J&J vaccine: did you mean F or to have some minus signs in front of the 2 and the 8? Where I live, it happens to be 8C today, and I just enjoyed brunch on the outside, socially distanced patio, wearing a jacket, unzipped.